
Compassionate Care at cCARE: What Patients Should Know
At cCARE, we are proud to provide trusted cancer care in our community. May is a time for awareness, education, and prevention. Learn more about our approach by reading our latest blog.
Monday, January 19, 2026 – Closed
In observance of Martin Luther King Jr. Day.
On December 11, 2020, the U.S. Food and Drug Administration (FDA) issued an Emergency Use Authorization (EUA) for the Pfizer-BioNtech mRNA vaccine for use against the virus that is causing the illness known as COVID-19. The vaccine has been 95% effective in large-scale clinical trials and has already been approved for use in five other countries. It is given in two doses, three weeks apart. The state of California is now administering vaccines to those 65 and older.
Another vaccine for the prevention of coronavirus disease 2019, this one from manufacturer Moderna called mRNA-1273, received EUA approval on December 18. Its efficacy rate is reported to be 94%. It is given in two doses, four weeks apart.
Both vaccines are different from other vaccines (such as for the flu or small pox) because they use a new technology called mRNA (messenger ribonucleic acid). That means these inoculations do not inject into the body an inactive or weakened sampling of the SARS-CoV-2 virus that causes the illness known as COVID-19. Rather, mRNA vaccines “teach” human cells how to make a chemical called a spike protein, which is the same protein found on the surface of the SARS-CoV-2 virus.
Once the immune system recognizes the “fake” protein, it triggers an immune response against the virus and starts making antibodies to fight it. The body can be protected against the virus without ever having to risk the potentially serious consequences of developing the COVID-19 illness.
After carefully studying all information and evidence, the doctors at cCARE say that the nonviral exposure vaccines (i.e., mRNA-based vaccines) should be taken by cancer patients. Potential side effects other than those already reported are not expected in cancer patients. The only potential issue is the possibility of a suboptimal antibody production response. The second dose (booster) would be an aid for a better response.
However, we do not recommend using the vaccine products that use a live or attenuated virus exposure. Currently the FDA has not issued an EUA for any of these types of vaccines. Patients currently receiving cancer treatment should discuss timing for use of the mRNA-based vaccine with their oncologist.
According to Centers for Disease Control and Prevention (CDC) guidelines, frontline healthcare workers and residents of long-term care facilities will be the first to be inoculated in Phase 1a. But the final decision on priority is determined by each state, either the governor or established advisory groups, as is the case in California.
While the majority of United States citizens may not have access to the vaccine until months into 2021, it is important for cancer patients and caregivers to start thinking about whether the vaccine is a good choice for those with cancer.
Cancer patients and those with a history of cancer are not included the CDC’s recommendation for the first group to receive the vaccine. However, adults with certain underlying medical conditions, including cancer, are at increased risk for the more severe forms of illness from the virus that causes COVID-19.
To that end, the California Department of Public Health recommends that adults aged 75 and older should be second in line (Phase 1b) to get the vaccine. Adults between the ages of 65-74 and those under age 64 who have underlying medical conditions fall under Phase 1c.
Initial studies that researched the efficacy of the COVID-19 vaccines did not include people undergoing cancer treatments or anyone who may have an otherwise weakened immune system. Therefore , it is not clear yet how effective the vaccines might be for these groups of people.
The American Cancer Society generally recommends that vaccines (other than the flu shot) not be given to patients undergoing chemotherapy, radiation treatments or immunotherapy. The reason is that, in order to work, a vaccine requires a healthy immune system response. Often cancer treatment suppresses the body’s immune system.
Keep in mind that most vaccines contain a small amount of live or inactive viruses. A healthy human body’s immune system response is to create antibodies to fight these viruses for the long term. The COVID-19 mRNA types of vaccine, however, do not contain the live virus that causes COVID-19.
Every cancer patient and cancer survivor’s situation is unique. We highly recommend speaking with your cancer doctor about the risks and benefits of getting the COVID-19 vaccine.
The two available vaccines do not contain an inactive or weakened sampling of the SARS-CoV-2 virus that causes the illness known as COVID-19. So caregivers who receive the vaccine are not at risk for passing the virus to the patient.
A caregiver should speak with his or her own doctor about whether the vaccine is right for them.
One important note is that it’s not clear yet whether a vaccinated adult who is later exposed to the virus can transmit the virus to someone else, even if he or she doesn’t get sick. Therefore, it is advised that those who get the vaccine continue to wear a mask and practice social distancing and good hand hygiene.
Very few serious safety concerns have been reported for the COVID-19 vaccines. The vaccines are still very new, and possible long-term side effects will continue to be studied.
The most serious, but rare, risk so far is an allergic reaction. Sometimes the ingredients in the vaccine on rare occasions can cause allergic reactions similar to those caused by allergies to bee stings and peanut butter. Reactions can include anaphylaxis, a severe condition that can cause shock, a rapid pulse rate, difficulty breathing, nausea and vomiting. Anyone receiving the COVID-19 vaccine should be monitored for 15 to 30 minutes after receiving the shot to mediate any complications.
Regardless of whether you get the COVID-19 vaccine, it is imperative to continue wearing a mask and social distancing. Stay in touch with your doctor to learn when the COVID-19 vaccine will become available in your area.
The best medicine for cancer patients, survivors and caregivers is to continue following state-mandated orders and CDC recommendations to protect themselves and others from COVID-19.
If at any time you have questions about COVID-19 symptoms or prevention, contact your healthcare provider.
The content provided here is for general information only and should not be relied upon or used as a substitute for professional medical advice, diagnosis or treatment. Any reliance on this information is at the risk of each website visitor. Always seek the advice of a physician or other qualified health provider with any questions regarding a medical condition.

At cCARE, we are proud to provide trusted cancer care in our community. May is a time for awareness, education, and prevention. Learn more about our approach by reading our latest blog.

Follow-up appointments are an important part of staying healthy. Whether you’re managing a condition, monitoring progress, or simply keeping up with routine care, these visits help your healthcare team make sure everything is on track.

Cancer treatment can bring physical, emotional, and mental challenges. Feeling stressed or overwhelmed is completely normal, but there are simple, effective ways to help manage stress and maintain a sense of control during this time.

At cCARE, we are proud to provide trusted cancer care in our community. May is a time for awareness, education, and prevention. Learn more about our approach by reading our latest blog.

Follow-up appointments are an important part of staying healthy. Whether you’re managing a condition, monitoring progress, or simply keeping up with routine care, these visits help your healthcare team make sure everything is on track.

Cancer treatment can bring physical, emotional, and mental challenges. Feeling stressed or overwhelmed is completely normal, but there are simple, effective ways to help manage stress and maintain a sense of control during this time.
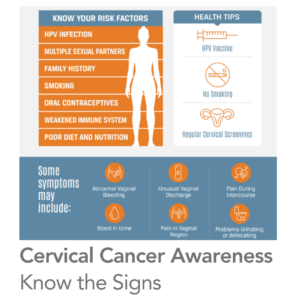
In early stages, cervical cancer may not cause noticeable symptoms. As it progresses, some signs may include: